The Rise to Quality Excellence
Bolstered by the recent acquisition of ISO 13485:2016 certification, Microbot Medical Inc. (NASDAQ: MBOT) has solidified its dedication to excellence in crafting the LIBERTY Endovascular Robotic Surgical System. This pivotal certification symbolizes the company’s march towards commercialization and regulatory adherence in key markets like the European Union and the United States.
With this approval in hand, Microbot Medical now stands empowered to forge ahead towards securing CE mark approval, paving the path for smoother navigation through the updated FDA regulations. This achievement acts as a beacon, guiding the company towards future growth and success in crucial markets.
The Game-Changing LIBERTY System
Microbot Medical, a pioneering force in the clinical-stage medical device realm, is focused on revolutionizing the landscape with its innovative micro-robotic technologies. The LIBERTY Endovascular Robotic Surgical System, the crown jewel of their endeavors, promises to redefine endovascular procedures by eliminating the need for bulky machinery, reducing radiation exposure, and lessening physician fatigue. Designed for potential remote operation, the LIBERTY system strives to democratize endovascular interventions, making them more accessible and efficient for all.
Viewing Beyond the Certification
The ISO 13485 certification serves as a validation of the company’s robust quality management system. This milestone isn’t just a feather in Microbot Medical’s cap; it’s a testament to the stringent standards they uphold in product development and manufacturing. Not only does this certification showcase their unwavering commitment to quality, but it also aligns them with the rigorous regulatory frameworks set forth by the European Union’s Medical Device Regulation and the FDA’s updated Quality System Management Regulation.
Exploring the Lucrative Future
Industry projections paint a bright future, with the robotic-assisted endovascular systems market anticipated to spike from $94 million in 2023 to a whopping $214.7 million by 2033, boasting a solid CAGR of 8.6%. This growth surge will be primarily fueled by the increasing adoption of cutting-edge systems that enhance procedural efficiency and precision, alongside the mounting demand for innovative, minimally invasive surgeries that cut down on patient trauma and recovery times.
Recent Strides with the LIBERTY System
In a recent significant stride, Microbot Medical shook hands with Emory University to explore potential collaboration in the realm of autonomous robotics within endovascular procedures. This partnership aims to birth a cutting-edge autonomous robotic system through the integration of the LIBERTY Endovascular Robotic Surgical System with an imaging system. The goal? A synergy of CT guidance, artificial intelligence, and medical robotics to uplift procedural standardization, efficiency, and patient accessibility.
This month, Microbot Medical bagged Institutional Review Board approval and inked a clinical trial agreement with Memorial Sloan Kettering Cancer Center (MSKCC) in New York City for the LIBERTY Endovascular Robotic Surgical System. MSKCC, powered by an Investigational Device Exemption, will steer the clinical trial to support upcoming FDA marketing submissions and commercialization. The trial spans across Brigham and Women’s Hospital and Baptist Hospital of Miami, already underway with conducting clinical cases.
Adding to the laurels, Microbot Medical recently disclosed that Baptist Hospital of Miami, alongside Miami Cardiac & Vascular Institute and Miami Cancer Institute, successfully conducted the LIBERTY Endovascular Robotic Surgical System’s maiden clinical procedure. This development came hot on the heels of Baptist Hospital joining the esteemed cohort of clinical trial sites and marks the second site to conduct such procedures, following Brigham & Women’s Hospital. This clinical trial under the Investigational Device Exemption for LIBERTY holds the promise of furnishing results crucial for future FDA submissions and commercialization.
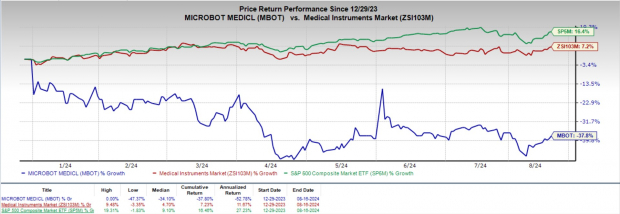
Price Performance and Strategic Outlook
Despite an overall industry upswing, Microbot Medical’s shares have taken a slight dip of 37.8% this year, against a 7.2% industry uptick and a 16.4% rise in the S&P 500 index.

Image Source: Zacks Investment Research
Assessing Zacks Rank & Top Contenders
Microbot Medical currently bears a Zacks Rank of #3 (Hold). However, shrewd investors eyeing the medical arena might find allure in stocks like Universal Health Services, Quest Diagnostics, and ABM Industries. Universal Health Services boasts a Zacks Rank of #1 (Strong Buy), while Quest Diagnostics and ABM Industries sport a Zacks Rank of #2 (Buy) each.
Universal Health Services anticipates a long-term growth rate of 19%, with earnings consistently surpassing estimates over the past four quarters, clocking an average of 14.58% above the predicted figures.
Quest Diagnostics, projecting a long-term growth rate of 6.20%, keeps the streak of surpassing earnings estimates alive over the previous four quarters, with an average surprise of 3.31%.
ABM Industries also dazzles with earnings eclipsing estimates in four consecutive quarters, delighting investors with an average surprise of 7.34%.
As the year unfolded, Universal Health Services witnessed a commendable 41.1% increase in shares, trumping the industry surge of 34.8%. Quest Diagnostics posted a 3.7% gain, while ABM Industries soared by 24.1%, outshining the industry’s 11.9% leap.